 Substances that ionize in solution are called electrolytes those that do not ionize, like sugar and alcohol, are called nonelectrolytes. Compounds that contain dissociable protons, or hydrogen ions, H +, or basic ions such as hydroxide ion, OH −, make acidic or basic solutions when they dissociate in water (see acids and bases dissociation). Compounds such as salts dissociate in solution into their ions, e.g., in solution sodium chloride exists as free Na + and Cl − ions. Ionization of neutral atoms can occur in several different ways. In the salt sodium carbonate, Na 2CO 3, two sodium cations are needed to neutralize each carbonate anion, CO 3 −2, because its charge is twice that of the sodium ion. In common table salt, or sodium chloride, NaCl, the sodium cations, Na +, are neutralized by chlorine anions, Cl −. Since ordinary matter is electrically neutral, ions normally exist as groups of cations and anions such that the sum total of positive and negative charges is zero. If an atom or group gains electrons or loses protons, it will have a net negative charge and is called an anion. If an atom or group loses electrons or gains protons, it will have a net positive charge and is called a cation. A simple ion consists of only one charged atom a complex ion consists of an aggregate of atoms with a net charge. Since the electron and proton have equal but opposite unit charges, the charge of an ion is always expressed as a whole number of unit charges and is either positive or negative. Positive and Negative Electric ChargesĪ neutral atom or group of atoms becomes an ion by gaining or losing one or more electrons or protons. 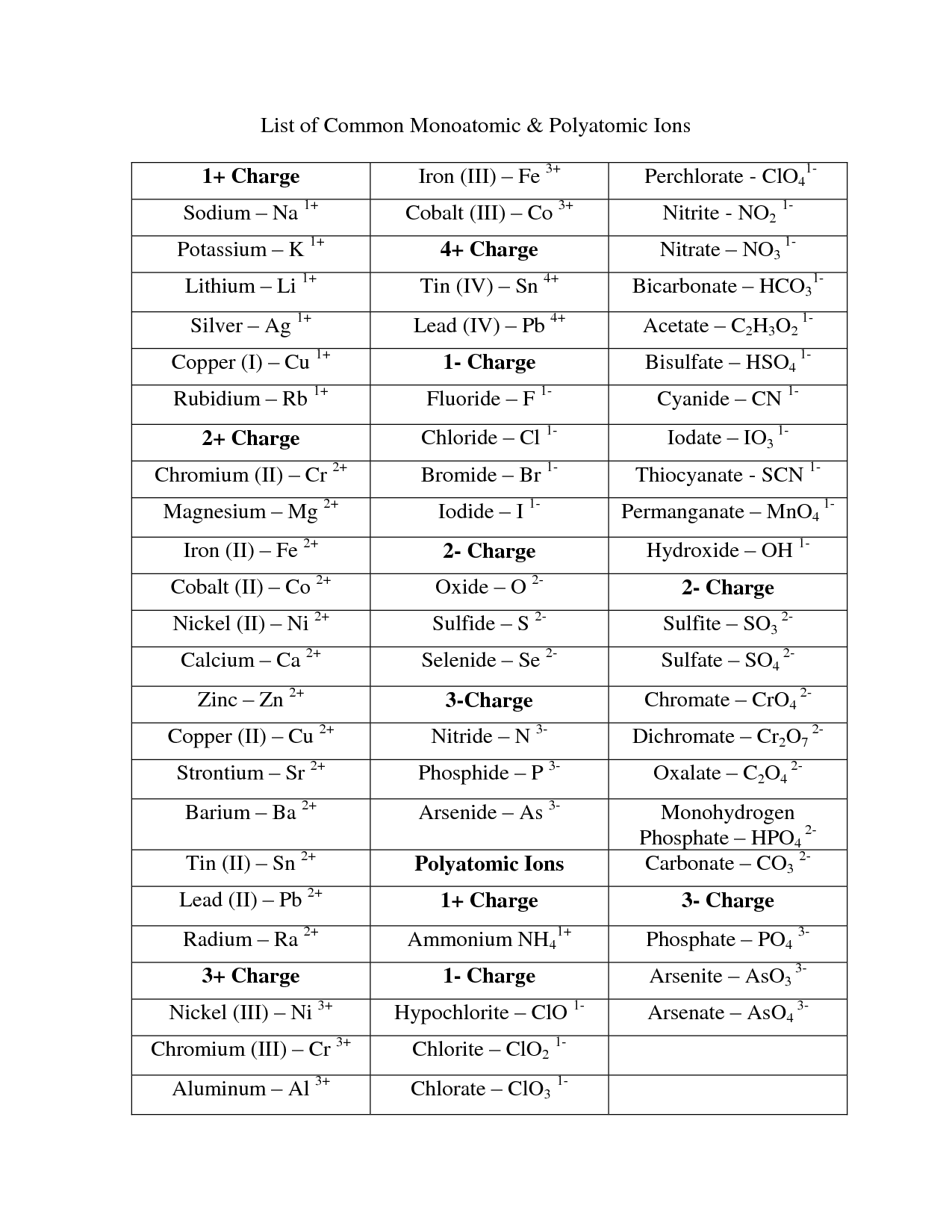
Ion, atom or group of atoms having a net electric charge. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
